PD Dr. Richard Carr
Experimental Pain Research, Medical Faculty Mannheim, Heidelberg University
Dr. Beatrice Oehler
Specialist for Anaesthesiology Group leader TRiP – Translational Research in Pain
Interactions between nociceptive and non-nociceptive circuits: intranasal
lidocaine to treat primary and secondary headache (human-rodent tandem project)
Background:
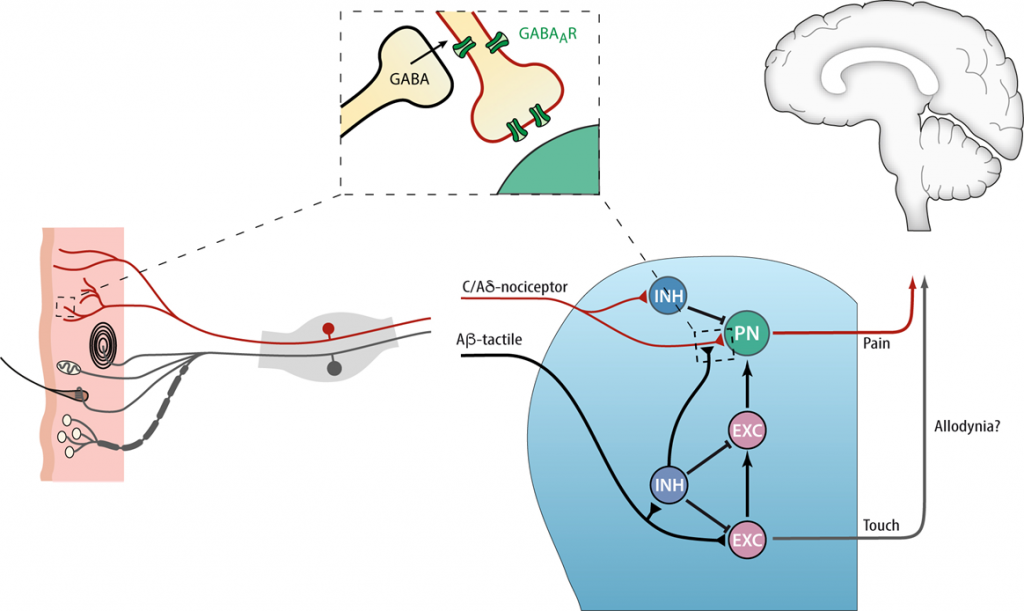 The relief of acute pain that we experience through the instinctive rubbing a freshly bruised shin bone occurs in the spinal dorsal horn through touch mediated inhibition of pain circuits. It has been suggested that disruption of spinal inhibition may exacerbate the development of post-injury pain. However, whether presynaptic inhibition onto primary afferents and/or post-synaptic inhibitory connections onto spinal cord neurons are dysregulated remains unresolved. Recently, we provided in vivoevidence for a presynaptic contribution by deleting the GABAAβ3 subunit selectively in peripheral nociceptive axons. This resulted in an increased basal sensitivity to acute thermal and mechanical pain and a failure to develop heat hyperalgesia after chronic constriction injury (CCI) and no signs of tactile allodynia following inflammatory Complete Freund’s Adjuvant (CFA). This implies that presynaptic inhibition differentially affects heat and mechanical sensory modalities dependent upon the type of injury.
The relief of acute pain that we experience through the instinctive rubbing a freshly bruised shin bone occurs in the spinal dorsal horn through touch mediated inhibition of pain circuits. It has been suggested that disruption of spinal inhibition may exacerbate the development of post-injury pain. However, whether presynaptic inhibition onto primary afferents and/or post-synaptic inhibitory connections onto spinal cord neurons are dysregulated remains unresolved. Recently, we provided in vivoevidence for a presynaptic contribution by deleting the GABAAβ3 subunit selectively in peripheral nociceptive axons. This resulted in an increased basal sensitivity to acute thermal and mechanical pain and a failure to develop heat hyperalgesia after chronic constriction injury (CCI) and no signs of tactile allodynia following inflammatory Complete Freund’s Adjuvant (CFA). This implies that presynaptic inhibition differentially affects heat and mechanical sensory modalities dependent upon the type of injury.
The project will use genetic techniques to target subsets of primary afferent neurons and GABAergic interneurons in the spinal cord.Cre lines combined with flex-excision (FLEX) viruseswill be used to map the structural architecture of micro-circuits involved in spinal pre-synaptic inhibition. Monosynaptic rabies virus-based tracing will identifyprojection targets of inhibitory interneurons while labelled GABAergic interneurons will be profiled functionally with patch-clamp and optogenetics in spinal cord slices. Viral techniques will also be used to render primary afferent populations photoactivatable (ChR2). Axonal threshold tracking and single fibre techniques will enable us to specify which functional classes of primary afferent Aδand C-fibre nociceptors are amenable to pre-synaptic modulation by virtue of their expression of functional axonal GABAAreceptors. In silico modelling will complement the electrophysiology to explore the functional impact of focal GABA in nociceptive nerve terminals. Behavioural testing will be used to probe causal relationships between diminished spinal presynaptic inhibition and the development of mechanical and thermal hyperalgesia following inflammation and nerve injury.
We aim to identify micro-circuits mediating interactions between non-nociceptive and nociceptive signals in the spinal dorsal horn with a focus on circuits involving presynaptic inhibitory contacts onto the central terminals of primary nociceptor afferents. This represents a key step towards delineating the impact of presynaptic inhibition on nociceptive processing following injury.



















